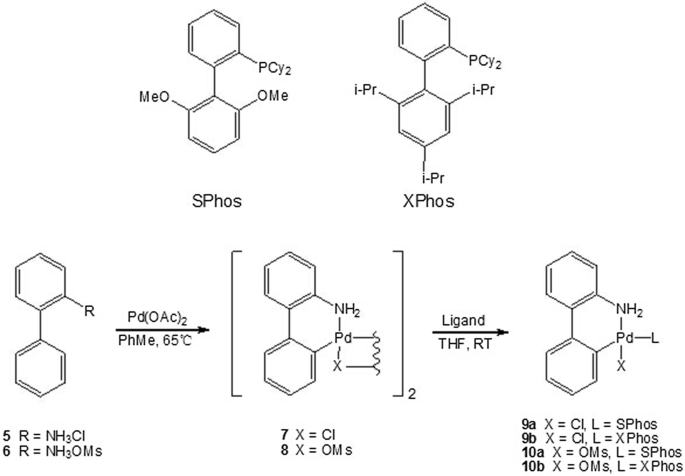
A new palladium precatalyst allows for the fast Suzuki-Miyaura coupling reactions of unstable polyfluorophenyl and 2-heteroaryl boronic acids. | Semantic Scholar
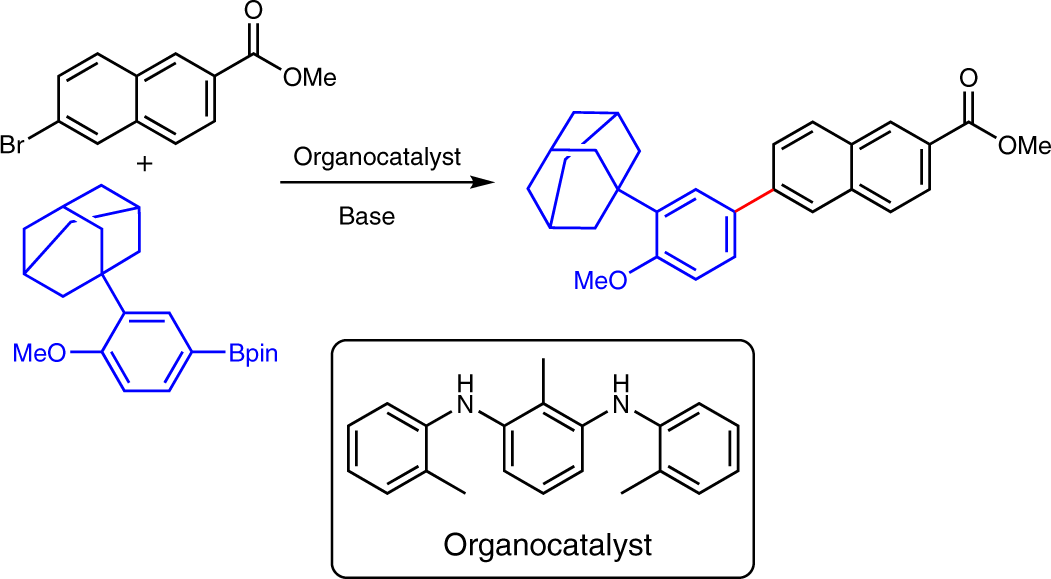
RETRACTED ARTICLE: The amine-catalysed Suzuki–Miyaura-type coupling of aryl halides and arylboronic acids | Nature Catalysis

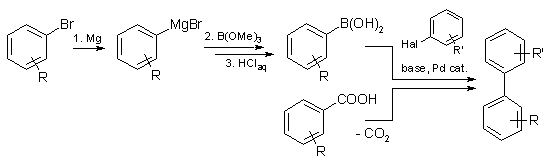
Synthesis of Biaryls via Decarbonylative Palladium-Catalyzed Suzuki-Miyaura Cross-Coupling of Carboxylic Acids - ScienceDirect
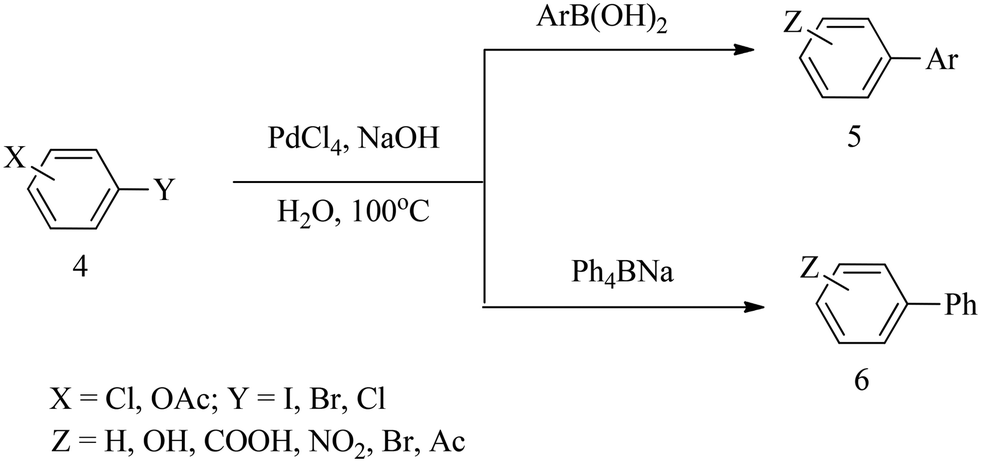
Recent advances in transition metal-catalysed cross-coupling of (hetero)aryl halides and analogues under ligand-free conditions - Catalysis Science & Technology (RSC Publishing) DOI:10.1039/C9CY01331H

Suzuki-Miyaura cross-coupling reaction of aryl chlorides with aryl boronic acids catalyzed by a palladium dichloride adduct of N-diphenylphosphanyl-2-aminopyridine - ScienceDirect

Palladium-Catalyzed, Direct Boronic Acid Synthesis from Aryl Chlorides: A Simplified Route to Diverse Boronate Ester Derivatives | Journal of the American Chemical Society

Regio- and stereoselective synthesis of tetra- and triarylethenes by N-methylimidodiacetyl boron-directed palladium-catalysed three-component coupling | Communications Chemistry

Palladium‐Catalyzed Synthesis of (Hetero)Aryl Alkyl Sulfones from (Hetero)Aryl Boronic Acids, Unactivated Alkyl Halides, and Potassium Metabisulfite - Shavnya - 2015 - Angewandte Chemie International Edition - Wiley Online Library
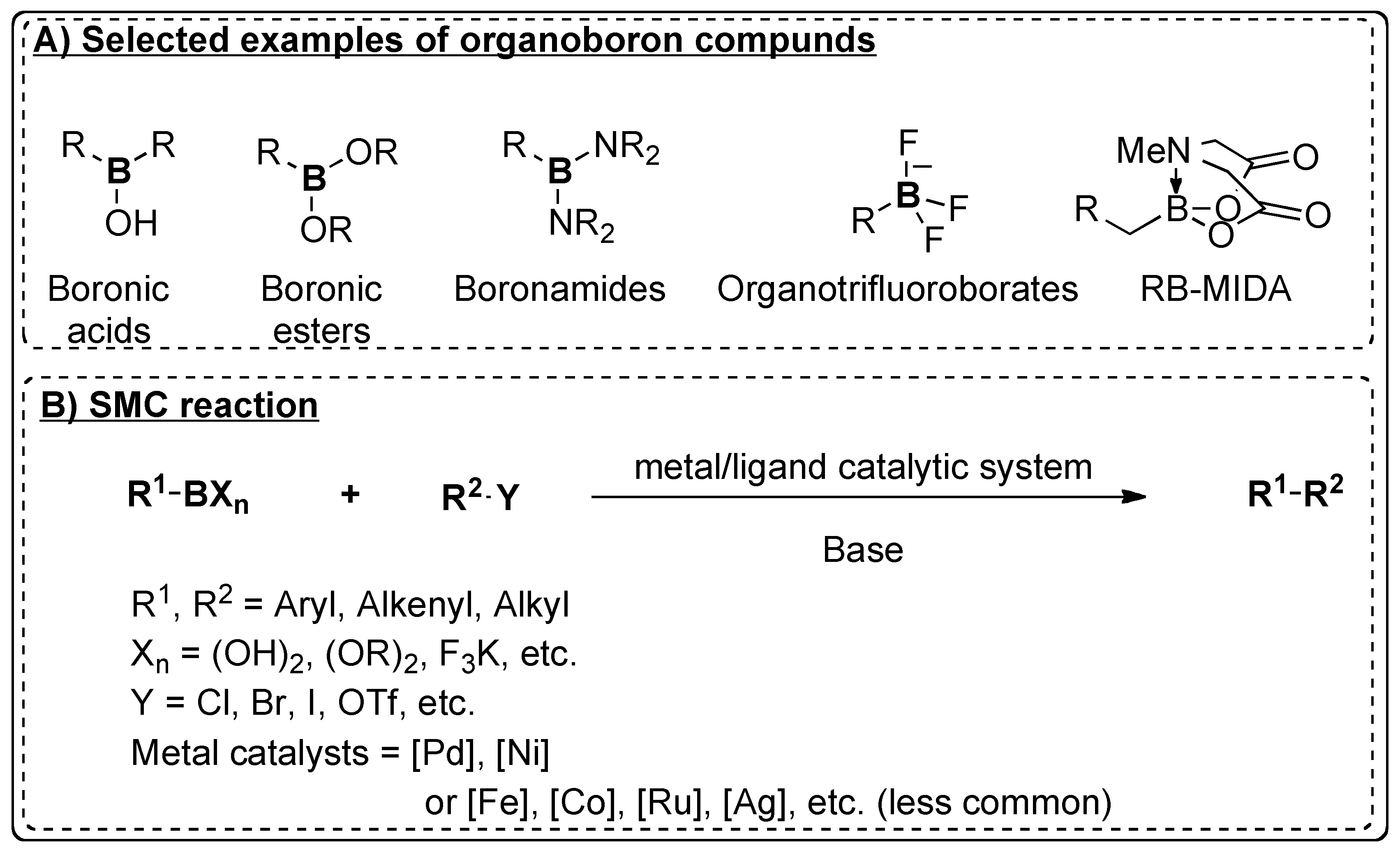
Catalysts | Free Full-Text | Recent Advances in Metal-Catalyzed Alkyl–Boron (C(sp3)–C(sp2)) Suzuki-Miyaura Cross-Couplings

Comparison between reactions. (a) aryl halide (1.1 mmol), arylboronic... | Download Scientific Diagram
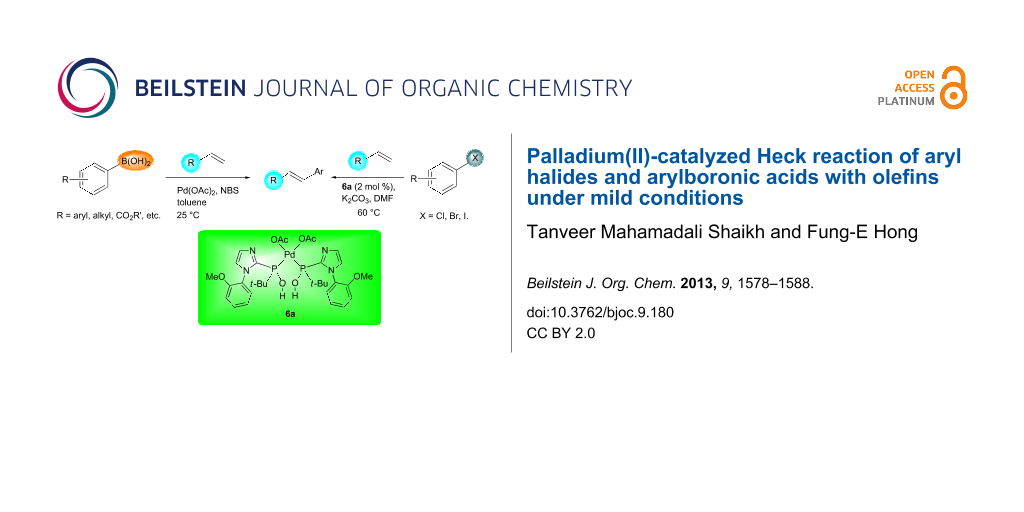
BJOC - Palladium(II)-catalyzed Heck reaction of aryl halides and arylboronic acids with olefins under mild conditions
![PDF] Room-temperature palladium-catalysed Suzuki–Miyaura coupling of arylboric acid with aryl chlorides | Semantic Scholar PDF] Room-temperature palladium-catalysed Suzuki–Miyaura coupling of arylboric acid with aryl chlorides | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/ddca6ac6a0773a85249c99e02c2f39093ca4d16e/3-Table1-1.png)
PDF] Room-temperature palladium-catalysed Suzuki–Miyaura coupling of arylboric acid with aryl chlorides | Semantic Scholar

Suzuki-Miyaura cross-coupling of phenylboronic acid with aryl halides catalyzed by palladium and nickel species supported on alumina-based oxides - ScienceDirect

Coupling of Aryl Halides with Aryl Boronic Acids with P(C6H5)(2-C6H4Cl)2 as the Supporting Ligand | Organometallics